|
Warm up today was a review of density and took a look at the difference between the density of salt water and fresh water. Next, we went over our homework on density. Here are the answers frfom last night's homework. Next, we finished our notes on page 19 on physical and chemical changes and notes on the back of page 19 about chemical reactions. Your can find the powerpoint here.
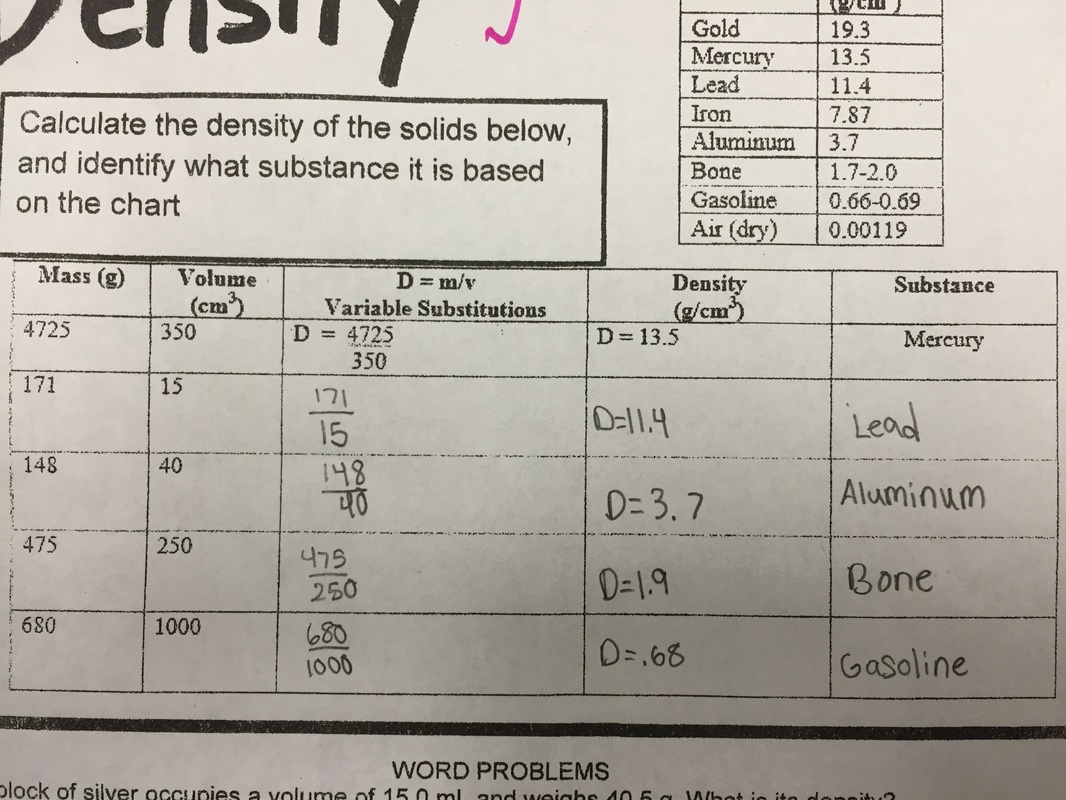
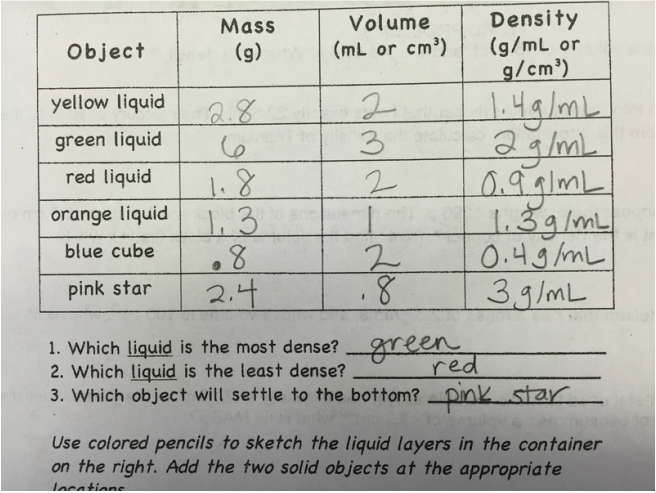
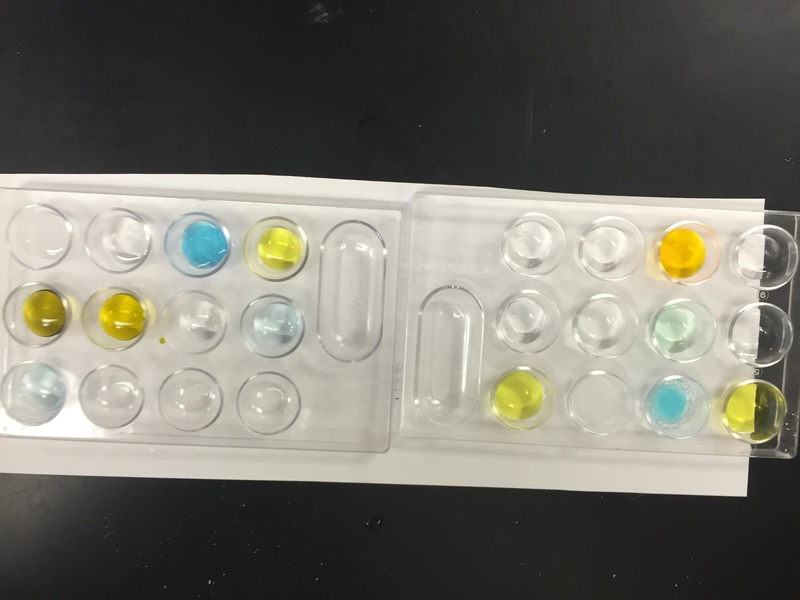
Finally, we conducted a lab utilizing physical and chemical properties and physical and chemical changes. Students identified some physical properties of 7 chemicals and then saw how the chemicals reacted. 1st block/A day---Tuesday morning we had an assembly first block, we will catch up what we missed today on Thursday, You still have homework tonight on density...due Thursday. Everyone else.... We did a density lab review as our warm up today. Just a way to practice calculating density (mass/volume)! Here are the conclusion questions for today's lab: If you put all of the liquids in a container, color a picture of how they would be layered. On your illustration write the average density for each liquid. Paragraph form •What did you learn from this lab? What can you tell me about density of a substance as the volume gets bigger? How do you calculate density? •If water has a density of 1.0g/mL , which liquids will float in water? sink in water? •What could have been some errors you made in the lab? •If we put water in the graduated cylinder you just drew, where would it go? Next, we graded our quizzes from last Thursday and Friday. The grades should be in Power Schools by this afternoon. If you did not take the quiz, you need to make arrangements to come before school or after school to take it. You will only need about 20 minutes to take the quiz. Lastly, your homework is a handout on density. Please have both sides completed by Thursday/Friday. I do not have an electronic copy of this assignment, and I don't have an extra copies.  Project due next Wednesday (A day) and Thursday (B day)! Quiz today! First we added notes on the back of page 18 in our notebooks on Ions and Isotopes. Next we took our quiz. We did a round about quiz. Students traveled around the room and answered the questions. Kind of an unusual way to take a quiz, but they did a GREAT job!!! Lastly, we took notes on page 22 in our on density. Here is the powerpoint for today.  Click here to vote for homecoming court memebers
We reviewed metals and non metals during our warm up today and then reviewed the groups on the periodic table from page 17 and 18 in our notebooks. Here is the powerpoint from today.
Quiz on THURSDAY will cover the following.
Reminder: Projects are due 10/12 - A day 10/13 - B Day Next, we did a quick lab about the Density of water and calculating the density of water. By doing the lab before the notes on density, students will be able to make inferences about density and calculating density. HW Pick 2 ELEMENTS from opposite sides of the periodic table. In paragraph form (2 paragraphs) …write as much information as possible about each element based on the periodic table. Include the following information for both elements: •Atomic number •Protons •Electrons •Neutrons •Atomic mass •Metal, non or metalloid •Period number •Group number •Reactivity-how reactive is it and what makes it act that way •Number of electron shells •Which other elements does your element have similarities with? Today we started the day with a review of Metals, nonmetals and metalloids by reviewing our conclusion from last class. Then, I assigned the the students project for 1st quarter. It is a choice project, they can pick one of the project to complete. The students were given a hard copy, but here is a digital copy of the directions. Projects are due Wednesday October 12th (A day) and Thursday October 13th (B Day). The goal for today is to introduce groups and periods on the periodic table. We took notes (page 17) on groups, periods, valence electrons, and reactivity. The students then used chrome books to watch four short videos on each of the groups on the periodic table. Here is the powerpoint for today's lesson. When using Discovery Education for this assignment, you do not need to be logged in to your rapid identity site. Just click on the link and type in your name. https://assignments.discoveryeducation.com/?cdPasscode=TE79E-44C0 IF YOU FINISH THE DISCOVERY ED ASSIGNMENT -click on the link for the Crash Course: Periodic Table video about Mendeleev and the history of the periodic table.
https://www.youtube.com/watch?v=0RRVV4Diomg When you are finished, click on the following link to take the quiz. https://docs.google.com/a/cms.k12.nc.us/forms/d/e/1FAIpQLSfLNCroce3HlwL9v178ZpH8KMuuduO5jhVUQuil5wo6tH31xA/viewform QUIZ today on Atoms and the periodic table. Grades should be loaded on Powerschools by the end of the day.
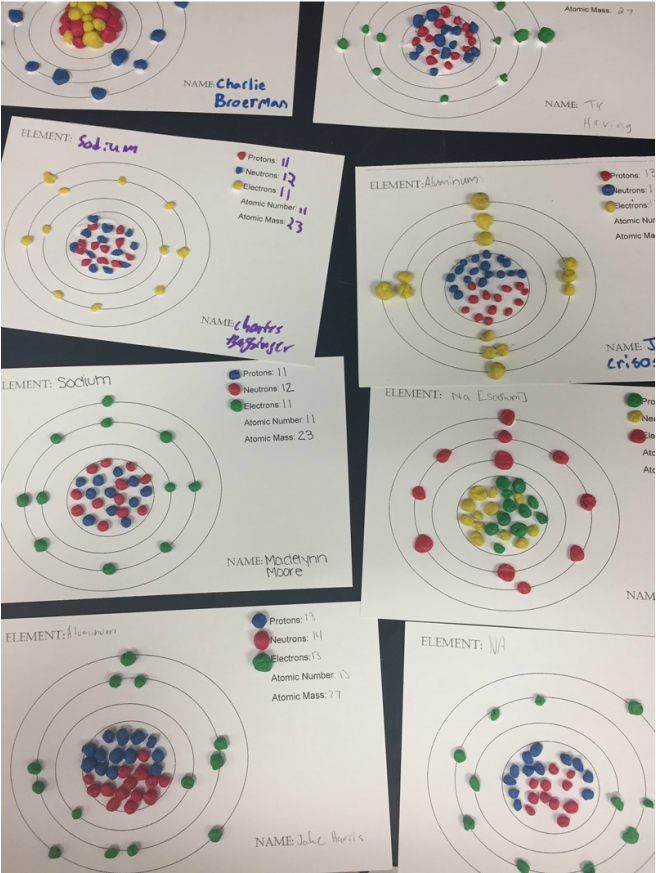
Next, we used the COACH books to read a short passage on synthetic and natural elements. This assignment was completed on page 15 in our notebooks. We then reviewed the differences between metals, non metals and metalloids, We added notes to our vocabulary pyramids from last class. Here is the powerpoint on the metal notes. We finished the day with a lab. Students were able to touch some elements and make observations about the physical properties of the elements. Students then used what they know about the differences between metals and nonmetals to determine if the elements were metals or nonmetals. Today we reviewed the parts of the atom and drawing the atoms. Next, we did a round robin around the room using the Atomic Structure Task Cards as a review of atomic structures. There will be a quiz on Wednesday/Thursday on reading the periodic table and drawing atoms. Make sure you have your periodic table in class. Lastly, we read a passage on metals, non metals and metalloids. Students created vocabulary pyramids for each of the vocabulary words and colored in a blank periodic table according to metals, non metals and metalloids. Here is the reading passage from today. Homework tonight is to take 8 atoms from last nights grid and draw the 8 atoms. Our main goal today was learning how to draw atoms from the elements on the periodic table. Here is the Powerpoint from today if you missed class. We practiced drawing several atoms, and each students created a model for a specific element. We also practiced our chemistry skills using the song below. Well done singers!!!
Homework tonight is attached to the back of page 11, it is a chart titled, "Reading the Periodic Table". Here is a copy of the handout. |
Archives
October 2016
Categories |
Photos used under Creative Commons from Nick Bastian Tempe, AZ, Cloud070













 RSS Feed
RSS Feed
